Relationship of Hip Muscle Performance to Leg Ankle and Foot Injuries a Systematic Review
- Systematic Review
- Open Access
- Published:
Are Leg Muscle, Tendon and Functional Characteristics Associated with Medial Tibial Stress Syndrome? A Systematic Review
Sports Medicine - Open book vii, Article number:71 (2021) Cite this article
Abstract
Background
Medial tibial stress syndrome (MTSS) is a common overuse injury that lacks constructive testify-based treatment options. Reduced leg girth has been associated with MTSS development considering information technology is hypothesised to impair the ability of the leg to attune tibial loading generated during pes–ground contact. Measuring total leg girth, however, does not provide specific information about the structural composition or functional capacity of individual leg muscles. Consequently, doubt remains every bit to which specific muscles are compromised and contribute to MTSS evolution. Therefore, this paper aimed to systematically review the body of literature pertaining to how the structure and part of the leg muscles are thought to be associated with MTSS injury.
Methods
The review was conducted post-obit the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols (PRISMA-P). Medline, PubMed, SCOPUS, SPORTDiscus with Full-texts and Web of Science were searched until March 2021 to identify articles in which lower limb muscle structural or functional variables associated with MTSS injury were investigated.
Results
Seventeen studies, which were predominately case–control in design and captured data from 332 individuals with MTSS symptoms and 694 command participants, were deemed appropriate for review. The average Downs and Black Quality Assessment score was 71.vii ± sixteen.4%, with these manufactures focussing on leg girth, tendon abnormalities, muscle strength and endurance, shear modulus and neuromuscular control. Of the hazard factors assessed in the 17 studies, decreased lean leg girth and college acme soleus muscle activity during propulsion were most strongly correlated with MTSS development. Individuals with MTSS also displayed deficits in ankle plantar flexor endurance, greater isokinetic concentric eversion strength, increased muscle shear modulus and contradistinct neuromuscular recruitment strategies compared to asymptomatic controls.
Conclusions
Future prospective studies are required to confirm whether decreased lean leg girth and higher top soleus muscle action during propulsion are associated with MTSS evolution and to elucidate whether these structural and functional differences in the leg muscles betwixt MTSS symptomatic and asymptomatic controls are a cause or effect of MTSS.
Key Points
-
Decreased lean leg girth is a likely risk cistron associated with developing MTSS, although this reduction in muscle girth is not related to the chapters of the leg muscles to produce maximal strength.
-
Higher acme soleus muscle activity during propulsion is likely associated with MTSS development.
-
The small number of prospective studies deemed appropriate to include in this review resulted in large knowledge gaps as to how leg muscle structure and function are associated with MTSS. Therefore, future prospective studies are necessary to assess leg muscle structural and functional characteristics in populations at run a risk of MTSS development.
Background
Running is a popular form of physical action primarily driven by the physical health benefits associated with exercise and the relative ease of accessibility to running [i]. Although the health benefits associated with running are widely acknowledged [ii], runners of all levels and disciplines feel overuse injuries, with those preparation for longer distances at greater take chances of injury [3]. Overuse injuries tin be problematic for runners because these injuries have the potential to disrupt active lifestyles and negatively influence the positive physiological adaptations gained from running [4].
1 overuse injury afflicting both novice and experienced runners is medial tibial stress syndrome (MTSS), more normally referred to using the outdated term, shin splints. With an incidence rate of between 4 and 35% [5,half dozen,seven], MTSS predominately affects individuals participating in activities that impose repetitive loading upon the lower limb, such every bit running [8]. Pain is diffuse, covering an area of at least five cm at the eye to distal third of the posteromedial tibial border [five]. MTSS is a separate pathology from a stress fracture, chronic exertional compartment syndrome and neuropathies affecting the lower limb [6].
To better inform MTSS treatment protocols, numerous research studies have been conducted to identify gamble factors for developing the injury. The authors of a systematic review and meta-analysis reported that the take chances factors for developing MTSS included female sex, previous history of MTSS, fewer years running experience, orthotic use, increased trunk mass index (BMI), increased navicular drop and greater hip external rotation range of motion in males [8]. Difficulty exists, however, in basing MTSS treatment protocols on these current hazard factors considering many of them cannot be hands modified. Furthermore, there is no high-quality evidence for the effect of whatsoever current intervention in managing MTSS based on these gamble factors [ix].
I take a chance cistron proposed to contribute to developing MTSS, which could be modified, but is yet to be extensively explored, is reduced leg girth [10]. Reduced leg musculature is thought to impair the ability of the leg to modulate tibial loading acquired by the ground reaction forces generated at foot–ground contact during the stance phase of running, resulting in increased tibial loading placing individuals at risk of developing MTSS [10]. Although leg girth provides a gross measure of muscular bulk, it does not provide detail as to the structural composition or functional capacity of the leg muscles. Therefore, we do not know which specific leg muscles might be compromised in individuals with MTSS or how this might impact lower limb role. Notwithstanding, leg muscle structure and function could be targeted and relatively hands modified with appropriate interventions to modulate tibial loading and reduce MTSS incidence.
Several research teams have assessed leg muscle structural and functional characteristics in individuals with and without MTSS [10,11,12,13,14,xv,16,17,18,19,xx,21,22,23,24]. To date, however, no publication could be located in which the outcomes of these studies were systematically reviewed or how changes to leg muscle structure and function might predispose individuals to MTSS injury. The long recovery catamenia from MTSS symptoms [25] combined with the high MTSS incidence charge per unit highlight that further exploration of modifiable chance factors for MTSS is needed to develop better treatment protocols. Therefore, this paper aimed to systematically review the body of literature pertaining to how the structure and function of the leg muscles were associated with MTSS injury. Our secondary aim was to develop recommendations to directly time to come inquiry studies to fill knowledge gaps related to leg structure and function with the ultimate goal of meliorate informing MTSS treatment protocols.
Methods
Literature Search Strategy
To conform to best practice guidelines for systematic literature reviews, nosotros conducted this review post-obit the Preferred Reporting Items for Systematic Reviews and Meta-Assay Protocols (PRISMA-P) (see Additional file one: Appendix 1 Prisma-P checklist). The review protocol was registered on the PROSPERO international prospective register for systematic reviews website (https://www.crd.york.ac.britain/prospero) (PROSPERO 2020 CRD42020154523). The question for this systematic literature review was 'Do individuals who develop and are afflicted with MTSS display differences in lower leg muscle structure or function compared to active asymptomatic individuals?'.
A systematic literature search was completed in March 2021, and relevant manufactures in which the authors had investigated leg muscle structure or function associated with MTSS injury were identified. This included all bachelor years through a series of systematic searches of the databases Medline, PubMed, SCOPUS, SPORTDiscus with Total-text and Web of Science (come across Fig. 1). The databases were searched past the lead author using combinations of the key search terms: (i) "medial tibial stress syndrome" OR "MTSS" OR "shin splints" OR "exertional medial tibial hurting"; (ii) "musc*", "calf" "lower*" and "leg" (see Additional file 2: Appendix ii-search strategy). The terms "musc*" and "lower*" were chosen to include every bit many articles as possible in which the authors investigated muscle, muscular or musculoskeletal structure or function and leg, lower limb and lower extremity, respectively.
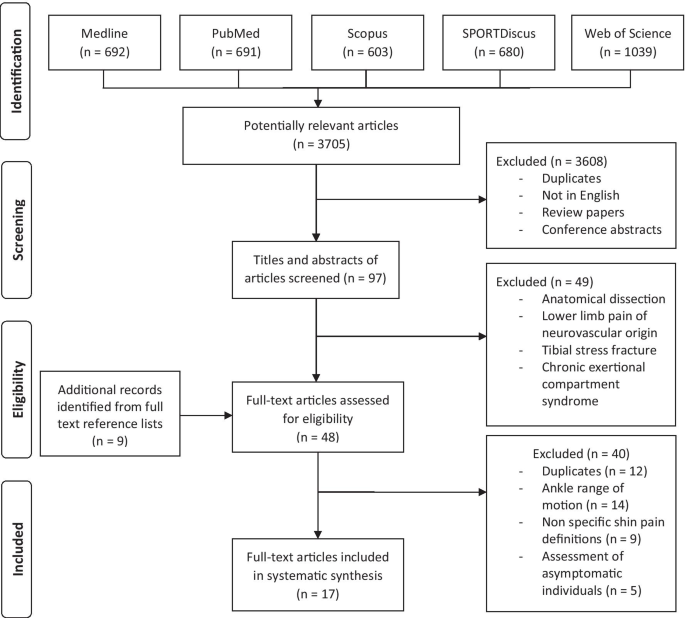
Systematic review menstruation diagram
Inclusion and Exclusion Criteria
In this systematic literature review, we divers MTSS according to the criteria described past Yates et al. [6]. All the same, studies that used alternating terminology to define MTSS were likewise included in the review if the study authors had excluded participants with a tibial stress fracture, chronic exertional compartment syndrome, lower leg tendinopathy and neurovascular pathologies.
Eligibility criteria were established before beginning the search. Articles were included if they were written in English and investigated lower limb muscle structural (excluding anatomical location) or functional variables associated with MTSS injury (e.g. a measure of leg circumference or ankle plantar flexor endurance). Additional relevant published papers were and then obtained from the reference lists of the sources located in the databases to help explicate and support the information presented throughout this review. Manufactures were excluded if the authors had investigated chronic exertional compartment syndrome, tibial stress fracture, lower leg tendinopathy or shin hurting of neurovascular origin.
Numerous authors accept assessed ankle joint plantar flexion and dorsiflexion range of motion associated with MTSS injury [half dozen, 26,27,28,29,thirty,31,32]. Although leg muscle structure and function can influence ankle joint range of motion, numerous other factors are too involved, such as the geometry of the articulating surfaces, the joint capsule, age and sex [33]. Equally this paper focussed on leg muscle structure and function, talocrural joint articulation range of motion was not included as an issue variable.
Procedures
The atomic number 82 author assessed the methodological quality of the studies described in the manufactures included in the review using the modified Downs and Black Quality Cess Checklist [34]. The Downs and Black Quality Assessment Checklist exhibits high test–retest and inter-rater reliability (r = 0.88 & 0.75) and criterion validity when compared with the global scores obtained using the Standards of Reporting Trials Grouping (r = 0.90) [34]. Therefore, the Downs and Black Quality Assessment Checklist is an appropriate tool to assess the methodological quality of both randomised and non-randomised health care intervention studies. The Quality Cess Checklist has recently been modified to allow a fairer appraisal of intervention and non-intervention studies [35, 36]. The Quality Cess Checklist reports on the bias of study reporting, internal and external validity and power. The following amendments to the Downs and Black Quality Assessment Checklist [34], modelled on the previous piece of work of Hebert-Losier et al. [35], were implemented for this review (come across Boosted file iii: Appendix 3-Modified Downs and Blackness Quality Cess Checklist). The terms 'patient' or 'subject' were replaced with 'participant' and 'treatment' interpreted in the context of testing. An boosted option of 'Not Applicable' was added to several questions (four, viii–ix, 12–14, 17, 19, 21–24, 26), which were deemed inappropriate to answer (i.e. the study was not an intervention study), and these questions were excluded from the total applicable points when this option was selected. Question 27 was simplified to comprehend statistical significance, whereby if a study reached statistical significance, information technology was answered 'Yes' (1 bespeak), and if it did non reach significance, information technology was answered 'No' (0 points). In Question xx, an commodity was scored 'Aye' if its author reported or referenced the level of accuracy of the instruments used in the study. When referencing confounders in Questions 5 and 25, age, sex, athletic action, competitive level or mensurate of weekly training and BMI were considered confounding variables. When assessing prospective studies, a history of MTSS was likewise considered a confounding variable. A score of 2 points was given if all principle confounders were reported. One point was awarded if 3 confounders were reported, and a score of zero was given when only 2 confounders were stated. All scores were then expressed, using Eq. (one), every bit a pct of the total applicable points.
$$\frac{\mathrm{Total\,number\,of\,points}}{\mathrm{Full\,applicable\,points}} \times 100$$
(1)
Two other authors reviewed controversial articles to reach a consensus before being included in the review. Once included in the review, data concerning the study design, aims, population (e.m. military or recreational athlete), muscle structural or functional variables, or interventions were assessed along with the implications for MTSS injury. Collated information were stored in a custom Microsoft Excel® 2016 spreadsheet (Microsoft Corporation, Redmond, WA, USA).
Statistical Analysis
Where relevant, data were pooled, and descriptive statistics were expressed as the pooled mean ± standard difference (see Eqs. 2 and 3) to summarise available data to assist in agreement the structural and functional characteristics of the leg muscles associated with MTSS. Additional data were sought from ane writer who had non responded to the asking at the time of publication.
$${\mathrm{Pooled}} {\bar{x}} =\frac{\left(\left({ {\bar{10}} }_{1}\times {n}_{1}\right)+\left({ {\bar{10}} }_{2}\times {north}_{two}\correct)+\cdots \left({ {\bar{x}} }_{x}\times {n}_{x}\right)\correct)}{ \Sigma n}$$
(two)
$$\mathrm{Pooled} \sigma =\frac{\left[\left({n}_{1}-1\right)\times {\sigma }_{i}\correct]+\left[\left({north}_{two}-1\right)\times {\sigma }_{2}\right]+\cdots \left[\left({northward}_{x}-ane\correct)\times {\sigma }_{10}\right]}{ \Sigma n-1x}$$
(3)
Results
The initial search results from five databases are shown in Fig. 1. After applying the inclusion criteria, 3705 studies remained. Later on the exclusion criteria were applied, 17 studies remained and were included in this review. Of the 17 articles, there were 13 case–control studies, 3 prospective accomplice studies and i example series. Using the modified Downs and Black Quality Assessment Checklist (run across Table i), the boilerplate score was 71.7 ± 16.4% (range 35–100%). The most common areas of poor operation with respect to the checklist were related to participants not existence representative of the source population and a lack of blinding to those measuring the master outcomes variables.
Inside this review, nine studies included participants from able-bodied populations of varying competitive levels, three studies included participants from military machine populations and five studies failed to study the study population, totalling 332 individuals with MTSS symptoms and 694 control participants (see Tabular array 2). The studies included in this review were predominately case–control in design. Therefore, the authors could not plant whether the characteristics associated with leg muscle structure or role were a cause or effect of MTSS. In the three prospective studies included in this review [10, 17, 37], the structural and functional characteristics of leg muscles were assessed and prospectively followed in asymptomatic armed forces and university athlete populations to determine whether these characteristics had implications for MTSS development. In full, 11 articles included assessments of functional characteristics [xi,12,thirteen, 15,16,17, 19, 21,22,23, 37] and 6 manufactures included assessments of structural characteristics [10, 14, 18, 20, 24, 38] of the leg muscles (encounter Tabular array 3). The assessed structural characteristics were lean and maximal leg girth and tendon construction [x, 14, 18, 20, 24, 38]. Pooled mean data from three case–control studies in which maximal leg girth was assessed [fourteen, xviii, 20] revealed no pregnant departure between MTSS symptomatic (n = 51; 379.2 ± 27.viii mm) and asymptomatic individuals (north = 177; 378.vii ± 31.four mm). The functional characteristics that were assessed were leg strength [11, 17, 21, 23], isotonic ankle plantar flexor endurance [12], leg musculus shear modulus [thirteen, 22] and neuromuscular control of leg muscles [15, 16, 19, 37]. The primary variables institute to be associated with MTSS included decreased lean leg girth and higher peak soleus muscle activity during propulsion and were deemed nigh likely to be associated with MTSS development based on the findings of the prospective studies. From example–control studies, decreased ankle plantar flexor endurance, greater isokinetic concentric eversion strength, increased leg muscle shear modulus and altered neuromuscular recruitment strategies were accounted possible adventure factors. Still, a causal relationship between the outcome variables identified from case–command studies and MTSS development cannot be determined (see Tabular array four).
Discussion
To our cognition, this is the first systematic review to critically assess the scientific literature pertaining to structural and functional leg muscle characteristics associated with MTSS. Despite numerous authors assessing gamble factors associated with MTSS evolution, in that location is a lack of loftier-quality evidence to determine whether individuals with MTSS brandish differences in leg muscle structure or function compared to agile asymptomatic individuals. Therefore, just 17 articles were accounted suitable to include within this systematic review. The primary variables found to exist associated with MTSS evolution in the 17 studies are discussed beneath.
Structural Characteristics
Leg Girth
Leg girth has been assessed equally a risk factor for developing MTSS because it is hypothesised that the amount of leg muscle bulk will influence the ability of the leg to attenuate ground reaction forces generated at foot–footing contact [10]. Researchers take used two protocols to mensurate leg girth: (i) lean leg girth and (ii) maximal leg girth. Lean leg girth is the maximal leg girth measured while a participant is continuing, corrected for adipose tissue thickness [10, 18]. Maximal leg girth is a standing not-corrected measure of leg girth [fourteen, xviii, 20].
In the only published prospective written report identified in this field, the report authors measured the maximal leg girth of 158 military recruits (122 men and 36 women). They constitute a statistically pregnant (p = 0.044) iv.2% reduction in the correct-sided lean leg girth of the 12 men who developed exertional medial tibial pain compared to the male person military machine recruits who did non develop the injury (command group) [10]. The diagnostic criteria for exertional medial tibial pain included an atraumatic seven-day history of at least ten cm of lengthened medial tibial pain. Although this definition differs from the current all-time practice injury definition [6], the diagnostic criteria fulfilled the key characteristics of the current all-time exercise MTSS injury definition and was therefore included in this review. Furthermore, although non statistically significant, men who developed exertional medial tibial pain had a hateful left lean leg girth that was 2.nine% less compared to the command group. Although there was no meaning difference between the lean leg girth of women who developed exertional medial tibial pain (due north = 11) compared to command participants (north = 25), the small number of women participants reduced the statistical ability of the report. In a case–command study of 15 MTSS symptomatic and 20 control male person military recruits [18], the authors did not find any significant departure between the 2 participant groups for lean leg girth.
Pooled mean information for maximal leg girth revealed no significant difference betwixt MTSS symptomatic and asymptomatic individuals. However, these pooled mean data should exist interpreted with caution due to limitations of the methodologies used to measure out maximal leg girth [xiv, 18, xx]. That is, Sobhani et al. [20] measured the maximal leg girth of the 181 armed services recruits merely to the nearest 0.5 cm, resulting in imprecise data. Furthermore, the case serial data presented by Clement [xiv] compared unilateral symptoms of MTSS individuals to the same individuals' non-afflicted limbs rather than to the limbs of matched control participants. Therefore, factors such as limb dominance and neuromuscular adaptations postal service-injury have the potential to confound these results.
Although but 1 study plant leg girth to be associated with developing MTSS, it was prospective in pattern, and it scored highest on the Quality Assessment Checklist of the studies in which leg girth was assessed [10]. The remaining studies in which leg girth was assessed were example–control in design. Therefore, the authors of these case–control studies were unable to ascertain whether the lack of difference in leg girth was an upshot of the injury or related to residue and subsequent morphological changes to the leg muscles. Information technology must as well exist acknowledged that circumferential measures of leg girth do not compensate for variances in bone volume and adipose tissue. Therefore, future research is warranted to quantify the relative proportion of lower limb muscle, bone and adipose tissue to determine how lean leg girth affects the composition of leg muscles in vivo to assist inform prevention strategies.
Leg Tendon Abnormalities
In combination with leg musculus limerick, lower limb tendon composition plays a pivotal role in attenuating ground reaction forces at foot–ground contact and transferring forces between leg muscles and bony structures to which they attach. In vivo assessment of the deep ankle plantar flexor tendons using musculoskeletal ultrasound of MTSS symptomatic and asymptomatic dancers has been provided by Winters et al. [38]. The authors assessed the flexor hallucis longus (FHL), flexor digitorum longus (FDL) and tibialis posterior (TP) of 15 MTSS symptomatic and 27 asymptomatic matched controls for pathological changes such as the presence of intratendinous and tendon sheath hypoechoic areas or hypoechoic oedema distending from the tendon sheath [38]. The authors reported that MTSS was not a role of tendon abnormality because both MTSS symptomatic and asymptomatic dancers displayed pathological changes within the tibialis posterior tendon [38].
Functional Characteristics
Leg Musculus Force and Endurance
Assessing leg structural characteristics provides surrogate measures upon which functional characteristics of the lower limb are inferred because they do not consider the functional capacity of the leg. To address this, several research teams [11,12,13, 15, 17, 19, 21,22,23] have assessed leg musculus strength and endurance, shear modulus and neuromuscular control metrics to better understand how leg musculus role differs between individuals with MTSS symptoms and asymptomatic individuals.
Researchers have investigated the association between MTSS and leg musculus strength and endurance to provide prove for the ii master theories associated with MTSS development: (i) muscular traction inducing periostitis [39,40,41,42] and (ii) a bone stress reaction of the tibial cortex associated with repetitive tibial loading and subsequent bending resulting in posteromedial tibial bony overload [5, 43,44,45,46]. To examination these theories, researchers have measured the strength of the muscles that most commonly attach to MTSS symptomatic locations and to quantify the relative forcefulness contribution of the leg muscles to decide their ability to modulate tibial loading. This review includes one case–control study in which isotonic ankle plantar flexor endurance was measured [12] and four case–control studies in which leg muscle force was measured using dynamometry [11, 17, 21, 23]. From the results of these studies, authors have fabricated inferences as to the potential role of these muscles in the development of MTSS.
Clinical rehabilitation protocols often comprise talocrural joint plantar flexion strengthening exercises into their treatment protocols [47]. The rationale for improving ankle plantar flexion endurance in MTSS symptomatic individuals is provided by Madeley et al. [12] who assessed 30 MTSS symptomatic individuals (median symptom duration xv weeks) and 30 sex, age and BMI matched controls. The authors reported that MTSS symptomatic individuals were able to complete significantly fewer single-leg heel raises (23 ± 5.6) compared to control participants (33 ± 8.6; p ≤ 0.001). Due to the potential influence of pain on the number of heel raises completed, hereafter prospective studies are required to determine whether the reduced ankle plantar flexion endurance capacity of MTSS symptomatic individuals was a cause or effect of MTSS.
Several authors have as well assessed the strength of leg muscles of individuals with MTSS using dynamometry. The studies by Hubbard et al. [17] and Saeki et al. [21] demonstrated that individuals who developed and had a history of MTSS displayed no statistically pregnant difference in the maximal voluntary isometric wrinkle (MVIC) strength of the leg plantar flexor, dorsiflexor, invertor or evertor musculus groups when compared to control individuals. Individuals with a history of MTSS, withal, displayed a significantly greater MVIC plantar flexion torque of the FHL (12.0 ± 3.0 Nm) compared to asymptomatic controls (9.viii ± 2.iii Nm; p = 0.04) [21]. However, Saeki et al. [21] concluded that the FHL is not likely to be related to the development of MTSS considering the FHL does non connect to the tibial fascia. Individuals with MTSS accept also been shown to display, on average, a significantly greater isokinetic concentric eversion strength (p < 0.05) [11], although there was no deviation in isokinetic dorsiflexion and plantar flexion strength compared to asymptomatic individuals [23]. Based on these results, the authors of the written report concluded that MTSS symptomatic individuals had a strength imbalance between the invertor and evertor muscles, whereby the evertor muscles were stronger [xi]. These findings should be interpreted with caution, however, considering age of activity initiation (p < 0.056) and grooming volume (p < 0.001) were significantly different between the two participant groups, with the control group starting time activeness at an earlier historic period and having a greater training volume than their counterparts with MTSS.
Current evidence suggests that individuals with MTSS accept a reduced isotonic ankle plantar flexor endurance capacity compared to asymptomatic matched controls. Nevertheless, these findings are not supported by a reduced strength of the ankle plantar flexor muscle group when assessed using dynamometry. Assessing the lower limb plantar flexor, dorsiflexor, invertor and evertor muscle groups equally a functional unit could mask variability in the strength of private muscles within that functional unit of measurement in MTSS symptomatic compared to asymptomatic controls. Potentially, individuals who are susceptible to MTSS could use compensatory musculus recruitment strategies to produce comparable force to those who exercise non develop MTSS. Therefore, future prospective research is warranted to assess ankle plantar flexor endurance and individual leg muscle strength to elucidate whether specific ankle plantar flexor muscles are responsible for the reduced isotonic ankle plantar flexor endurance capacity seen in individuals with MTSS. Furthermore, although current enquiry indicates that a strength imbalance between the invertor and evertor muscles is credible during isokinetic but non isometric contraction in individuals with MTSS, whether this is a cause or effect of MTSS requires farther prospective assessment.
Leg Musculus Shear Modulus
Although reduced ankle dorsiflexion range of movement is not reported to be a run a risk factor associated with MTSS development [eight, 9], MTSS symptomatic individuals commonly report a awareness of increased musculus tightness of the ankle joint plantar flexors [8]. Two case–control studies [thirteen, 22] were reviewed in which the inquiry teams used shear wave elastography to quantify leg muscle tightness in men, represented via the shear modulus of the muscles of the superficial posterior compartment of the leg (lateral gastrocnemius (LG), medial gastrocnemius (MG), soleus (SOL) and peroneus longus (PL)). Akiyama et al. [13] also assessed an antagonist to ankle plantar flexion, the tibialis anterior (TA), whereas Saeki et al. [22] specifically assessed the muscles contributing to ankle plantar flexion, and therefore also included FDL, FHL, peroneus brevis (Atomic number 82) and TP. Although both studies identified an increase in the shear modulus of several muscles in symptomatic participants relative to controls, at that place was a lack of consistency in the muscles identified between the studies. Akiyama et al. [13] ended that individuals with MTSS had a greater shear modulus of the LG, MG, PL, SOL and TA, whereas Saeki et al. [22] concluded that only the FDL and TP demonstrated an increase in shear modulus in individuals with MTSS compared to asymptomatic controls. These betwixt-study differences in results can be attributed to several factors, including different participant inclusion criteria. For example, Akiyama et al. [13] assessed individuals with MTSS who were symptomatic at the time of testing, whereas, in guild to avoid the confounding variable of pain, Saeki et al. [22] assessed individuals with a history of MTSS, excluding individuals with pain at the time of testing. Both research teams also used different metrics to written report the shear modulus, preventing the pooling of the data. Despite these limitations, conclusions drawn from the available data suggest an increase in shear modulus of the lower limb muscles, specially of the ankle plantar flexors, in MTSS symptomatic men that persists post-obit recovery of symptoms. For more meaningful conclusions to exist reached, future prospective research, including both men and women, is required to avoid the misreckoning variable of pain on shear modulus and make up one's mind whether a causal human relationship tin be established betwixt the shear modulus of the leg muscles and the development of MTSS.
Neuromuscular Control
Much of the previous research examining functional characteristics of the leg that might predispose individuals to MTSS has been limited by the experimental job being non-weight bearing or non activity specific for the individual (e.g. isometric or isokinetic muscle wrinkle) [eleven, 13, 17, 21,22,23]. One method to better sympathize the functional chapters of the leg muscles is to monitor the neuromuscular control of these muscles during dynamic tasks, such equally running or walking. One prospective and two example–control studies were located in which neuromuscular command of the leg muscles in individuals with MTSS and asymptomatic individuals were assessed [xv, 19, 37]. In a prospective report, Naderi et al. [37] assessed SOL and TA electromyographic (EMG) signals during the stance phase of running of 112 active university students. During a 17-week follow-up period, 23 (9 men and xiv women) individuals were diagnosed with MTSS. The authors constitute a statistically significant higher summit SOL EMG amplitude during assimilation (p = 0.01) and propulsion (p = 0.02) in the MTSS group compared to the control grouping, although no pregnant deviation was found for TA. Using a stepwise logistic regression analysis, the authors concluded that higher peak EMG amplitude of the SOL during propulsion significantly increased the risk of MTSS development by 5% (p = 0.01). The authors hypothesised that the increased SOL EMG aamplitude during propulsion could induce greater SOL traction on the posteromedial tibia, particularly in individuals with a higher dynamic foot posture due to greater and prolonged SOL contraction [37]. In a case–control comparison, Franettovich et al. [xv] assessed EMG signals of 12 lower limb muscles and lower limb kinematics while 14 female participants who had a history of exercise-related leg hurting within the previous 12 months and fourteen sex, age, superlative and weight-matched asymptomatic controls walked on a treadmill. Individuals with a history of practise-related leg hurting demonstrated a significant (p = 0.048) reduction in LG height activeness by xx.5% and 1.7% MVIC during stance and swing. Despite differences in meridian LG muscle activity, the authors observed no differences in foot posture, foot mobility or motion at the pelvis, hip, human knee and ankle betwixt the participant groups. Franettovich et al. [15] hypothesised that lower peak LG activity could potentially increase posteromedial tibial loading and MTSS development or exist a compensatory effect of exercise related leg pain symptoms. In addition, Rathleff et al. [19] assessed SOL and TA EMG signals along with midfoot kinematics from 14 MTSS symptomatic and 11 asymptomatic controls during treadmill walking. Rathleff et al. [19] reported that MTSS symptomatic individuals displayed a significant increment in complexity of the TA (p = 0.01) and SOL (p = 0.02) EMG signals with a lower complexity of midfoot kinematics compared to command participants. Complexity was defined as the caste of variability inside the betoken, where lower values indicated a more than regular bespeak and higher values indicated an increase in randomness in the signal [19]. Rathleff et al. [19] concluded that the increased complexity of the TA and SOL EMG signals and lower midfoot kinematic complication in the MTSS symptomatic group were associated with less motility variability. Increased complexity of EMG activity is hypothesised to be caused by the effects of adaptation due to pathology [48]. Contention remains, however, as to whether the neuromuscular system up or down-regulates in response to pathology [49, 50]. This finding supports the notion of Hamill et al. [51], who hypothesised that increased tissue stress in patellofemoral pain was caused by individuals who experienced a narrow range of movement with less movement variability compared to healthy controls. Lower limb muscle EMG signal adaptations reported past Naderi et al. [37], Franettovich et al. [fifteen] and Rathleff et al. [xix] build on the earlier work of Garth et al. [16], who demonstrated that individuals with posteromedial shin hurting displayed significant impairment of intrinsic muscles controlling the toe flexors and extensors compared to control participants. Furthermore, the findings by Franettovich et al. [15] and Rathleff et al. [19] are also consequent with previous research in which individuals with MTSS were able to produce force output that was comparable to asymptomatic individuals [17, 21], although possibly using compensatory muscle recruitment strategies to attain this.
Findings of the three studies included for review demonstrate that altered leg muscle recruitment strategies of the triceps surae are associated with MTSS injury. The findings of Naderi et al. [37] build on earlier work by Franettovich et al. [15] who assessed the SOL, LG and MG and concluded that when matched to controls, MTSS symptomatic individuals displayed lower peak LG musculus activation only. Edifice on the findings of Naderi et al. [37], it could be hypothesised that once individuals experience MTSS pain, they alter their neuromuscular patterning to reduce SOL muscle activation and associated muscular traction on the tibia as a pain management strategy. To substantiate this notion, all the same, farther prospective assessment of neuromuscular patterning of the triceps surae by individuals who develop MTSS is required.
Limitations
The primary limitation of this review is that most studies included for appraisement were instance–command in design, limiting our ability to determine whether the assessed factors were a cause or upshot of MTSS. In this review, we identified decreased lean leg girth as a likely take a chance factor associated with developing MTSS. We admit, however, that decreased lean leg girth does not account for variances in tibial bone volume. Furthermore, a lack of consistent testing procedures and issue variables in the studies we reviewed reduced our power to pool a greater proportion of data, limiting our ability to draw business firm conclusions.
Conclusions
The primal findings from the prospective studies included for review betoken that decreased lean leg girth is a probable run a risk cistron for developing MTSS [x], although this reduction in muscle girth was non related to the capacity of the leg muscles to produce maximal strength [17]. Furthermore, college peak soleus muscle activity during propulsion is associated with an increased risk of MTSS evolution [37]. Cross-sectional comparing of individuals suffering MTSS compared to asymptomatic controls demonstrated deficits in ankle plantar flexor endurance, greater isokinetic concentric eversion forcefulness, increased muscle shear modulus and contradistinct neuromuscular recruitment strategies. However, whether these differences between symptomatic patients and controls were due to pain or associated with injury development needs to exist further explored.
This review highlights large knowledge gaps in the bachelor literature investigating how leg musculus structure and part are associated with MTSS development. These knowledge gaps are due to the lack of prospective studies, which are required to identify a causal relationship between risk factors and MTSS development, and the failure of researchers to mitigate the misreckoning variable of pain. Future enquiry should assess specific structural characteristics of the leg, such as the relative proportion of in vivo lower limb musculus, os and adipose tissue, to decide whether specific muscles are responsible for the reduction in lean leg girth between individuals who develop MTSS compared to asymptomatic controls. Finally, future research should assess ankle plantar flexor endurance, individual leg muscle strength and the neuromuscular patterning of the triceps surae during running to meliorate inform MTSS injury prevention strategies and rehabilitation protocols.
Availability of Data and Material
All data generated or analysed during this study are included in this published article.
Code Availability
Not applicative.
Abbreviations
- BMI:
-
Body mass index
- EMG:
-
Electromyographic
- FDL:
-
Flexor digitorum longus
- FHL:
-
Flexor hallucis longus
- LG:
-
Lateral gastrocnemius
- MG:
-
Medial gastrocnemius
- MTSS:
-
Medial tibial stress syndrome
- MVIC:
-
Maximal voluntary isometric contraction
- PB:
-
Peroneus brevis
- PL:
-
Peroneus longus
- PRISMA-P:
-
Preferred reporting items for systematic reviews and meta-assay protocols
- SOL:
-
Soleus
- SD:
-
Standard departure
- TA:
-
Tibialis anterior
- TP:
-
Tibialis posterior
References
-
Kozlovskaia Yard, Vlahovich N, Rathbone Due east, Manzanero S, Keogh J, Hughes DC. A profile of health, lifestyle and training habits of 4720 Australian recreational runners—the case for promoting running for health benefits. Health Promot J Aust. 2019. https://doi.org/ten.1002/hpja.30.
-
Lavie CJ, Lee D, Sui X, Arena R, O'Keefe JH, Church TS, et al. Effects of running on chronic diseases and cardiovascular and all-cause mortality. Mayo Clin Proc. 2015. https://doi.org/x.1016/j.mayocp.2015.08.001.
-
Kluitenberg B, van Middelkoop G, Diercks R, van der Worp H. What are the differences in injury proportions between dissimilar populations of runners? A systematic review and meta-analysis. Sports Med. 2015. https://doi.org/ten.1007/s40279-015-0331-ten.
-
Van Middelkoop 1000, Kolkman J, Van Ochten J, Bierma-Zeinstra SM, Koes B. Prevalence and incidence of lower extremity injuries in male marathon runners. Scand J Med Sci Sports. 2008. https://doi.org/x.1111/j.1600-0838.2007.00683.x.
-
Moen MH, Tol JL, Weir A, Steunebrink M, De Winter TC. Medial tibial stress syndrome a critical review. Sports Med. 2009. https://doi.org/10.2165/00007256-200939070-00002.
-
Yates B, White S. The incidence and risk factors in the development of medial tibial stress syndrome among naval recruits. Am J Sport Med. 2004. https://doi.org/10.1177/0095399703258776.
-
Bennett JE, Reinking MF, Pluemer B, Pentel A, Seaton M, Killian C. Factors contributing to the development of medial tibial stress syndrome in high schoolhouse runners. J Orthop Sports Phys Ther. 2001. https://doi.org/x.2519/jospt.2001.31.nine.504.
-
Newman P, Witchalls J, Waddington Thousand, Adams R. Take a chance factors associated with medial tibial stress syndrome in runners: a systematic review and meta-analysis. Open up Access J Sports Med. 2013. https://doi.org/10.2147/oajsm.s39331.
-
Winters Grand, Eskes One thousand, Weir A, Moen MH, Backx FJG, Bakker EWP. Treatment of medial tibial stress syndrome: a systematic review. Sports Med. 2013. https://doi.org/10.1007/s40279-013-0087-0.
-
Burne SG, Khan KM, Boudville Atomic number 82, Mallet RJ, Newman PM, Steinman LJ, et al. Adventure factors associated with exertional medial tibial hurting: a 12 month prospective clinical study. Br J Sports Med. 2004. https://doi.org/10.1136/bjsm.2002.004499.
-
Yüksel O, Ozgurbuz C, Ergun K, Islegen C, Taskiran Eastward, Denerel N, et al. Inversion/eversion strength dysbalance in patients with medial tibial stress syndrome. J Sport Sci Med. 2011;10:737–42.
-
Madeley LT, Munteanu SE, Bonanno DR. Endurance of the talocrural joint joint plantar flexor muscles in athletes with medial tibial stress syndrome: a instance-control written report. J Sci Med Sport. 2007. https://doi.org/10.1016/j.jsams.2006.12.115.
-
Akiyama K, Akagi R, Hirayama G, Hirose N, Takahashi H, Fukubayshi T. Shear modulus of the lower leg muscles in patients with medial tibial stress syndrome. Ultrasound Med Biol. 2016. https://doi.org/10.1016/j.ultrasmedbio.2016.03.010.
-
Clement DB. Tibial stress syndrome in athletes. Am J Sport Med. 1974. https://doi.org/10.1177/036354657400200203.
-
Franettovich 1000, Chapman AR, Blanch P, Vicenzino B. Altered neuromuscular control in individuals with do-related leg pain. Med Sci Sports Exerc. 2010. https://doi.org/10.1249/MSS.0b013e3181b64c62.
-
Garth WP, Miller ST. Evaluation of claw toe deformity, weakness of the foot intrinsics and posteromedial shin pain. Am J Sport Med. 1989. https://doi.org/x.1177/036354658901700617.
-
Hubbard TJ, Carpenter EM, Cordova ML. Contributing factors to medial tibial stress syndrome: a prospective investigation. Med Sci Sports Exerc. 2009. https://doi.org/10.1249/MSS.0b013e31818b98e6.
-
Moen MH, Bongers T, Bakker EW, Zimmermann WO, Weir A, Tol JL, et al. Adventure factors and prognostic indicators for medial tibial stress syndrome. Scand J Med Sci Sports. 2012. https://doi.org/10.1111/j.1600-0838.2010.01144.ten.
-
Rathleff MS, Samani A, Olesen CG, Kersting UG, Madeleine P. Changed relationship between the complexity of midfoot kinematics and muscle activation in patients with medial tibial stress syndrome. J Electromyogr Kinesiol. 2011. https://doi.org/10.1016/j.jelekin.2011.03.001.
-
Sobhani V, Shakibaee A, Aghda AK, Meybodi MKE, Delavari A, Jahandideh D. Studying the relation betwixt medial tibial stress syndrome and anatomic and anthropometric characteristics of war machine male personnel. Asian J Sports Med. 2015. https://doi.org/x.5812/asjsm.23811.
-
Saeki J, Nakamura M, Nakao S, Fujita K, Yanase Thou, Morishita 1000, et al. Ankle and toe musculus strength characteristics in runners with a history of medial tibial stress syndrome. J Human foot Talocrural joint Res. 2017. https://doi.org/10.1186/s13047-017-0197-2.
-
Saeki J, Nakamura Thou, Nakao S, Fujita One thousand, Yanase K, Ichihashi N. Muscle stiffness of posterior lower leg in runners with a history of medial tibial stress syndrome. Scand J Med Sci Sports. 2018. https://doi.org/x.1111/sms.12862.
-
Ercan S. Ankle isokinetic muscle forcefulness and navicular driblet in athletes with medial tibial stress syndrome. Cukurova Med J. 2019. https://doi.org/10.17826/cumj.459411.
-
Sabeti Five, Khoshraftar YN, Bijeh North. The human relationship betwixt shin splints with anthropometric characteristics and some indicators of trunk limerick. J Sports Med Phys Fit. 2019. https://doi.org/10.23736/s0022-4707.16.05156-2.
-
Moen MH, Holtslag 50, Bakker E, Barten C, Weir A, Tol JL, et al. The handling of medial tibial stress syndrome in athletes; a randomized clinical trial. BMC Sports Sci Med Rehabil. 2012. https://doi.org/x.1186/1758-2555-four-12.
-
Andrish JT, Bergfeld JA, Walheim J. A prospective study on the management of shin splints. J Bone Joint Surg Am. 1974. https://doi.org/10.2106/00004623-197456080-00021.
-
Bartosik KE, Sitler M, Hillstrom HJ, Palamarchuk H, Huxel Thou, Kim E. Anatomical and biomechanical assessments of medial tibial stress syndrome. J Am Podiatr Med Assoc. 2010. https://doi.org/10.7547/1000121.
-
Becker J, James S, Wayner R, Osternig L, Chou LS. Biomechanical factors associated with achilles tendinopathy and medial tibial stress syndrome in runners. Am J Sport Med. 2017. https://doi.org/10.1177/0363546517708193.
-
Becker J, Nakajima M, Wu WFW. Factors contributing to medial tibial stress syndrome in runners: a prospective study. Med Sci Sports Exerc. 2018. https://doi.org/10.1249/mss.0000000000001674.
-
Garnock C, Witchalls J, Newman P. Predicting private risk for medial tibial stress syndrome in navy recruits. J Sci Med Sport. 2018. https://doi.org/x.1016/j.jsams.2017.10.020.
-
Loudon JK, Dolphino MR. Employ of foot orthoses and calf stretching for individuals with medial tibial stress syndrome. Foot Ankle Spec. 2010. https://doi.org/10.1177/1938640009355659.
-
Yagi S, Muneta T, Sekiya I. Incidence and take chances factors for medial tibial stress syndrome and tibial stress fracture in loftier school runners. Genu Surg Sports Traumatol Arthrosc. 2013. https://doi.org/x.1007/s00167-012-2160-x.
-
Grimston SK, Nigg BM, Hanley DA, Engsberg JR. Differences in talocrural joint articulation complex range of movement as a function of historic period. Pes Talocrural joint. 1993. https://doi.org/10.1177/107110079301400407.
-
Downs SH, Blackness North. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Commun Wellness. 1998. https://doi.org/10.1136/jech.52.6.377.
-
Hebert-Losier 1000, Supej M, Holmberg HC. Biomechanical factors influencing the performance of elite tall ski racers. Sports Med. 2014. https://doi.org/ten.1007/s40279-013-0132-z.
-
Forsyth JR, Riddiford-Harland DL, Whitting JW, Sheppard JM, Steele JR. Essential skills for superior moving ridge-riding performance: a systematic review. J Strength Cond Res. 2019. https://doi.org/10.1519/jsc.0000000000003402.
-
Naderi A, Moen MH, Degens H. Is high soleus muscle activity during the stance phase of the running cycle a potential hazard cistron for the development of medial tibial stress syndrome? A prospective study. J Sports Sci. 2020. https://doi.org/10.1080/02640414.2020.1785186.
-
Winters M, Bon P, Bijvoet Southward, Bakker EWP, Moen MH. Are ultrasonographic findings similar periosteal and tendinous edema associated with medial tibial stress syndrome? A case-control study. J Sci Med Sport. 2017. https://doi.org/10.1016/j.jsams.2016.07.001.
-
Stickley CD, Hetzler RK, Kimura IF, Lozanoff Southward. Crural fascia and muscle origins related to medial tibial stress syndrome symptom location. Med Sci Sports Exerc. 2009. https://doi.org/10.1249/MSS.0b013e3181a6519c.
-
Dark-brown AA. Medial tibial stress syndrome: muscles located at the site of hurting. Scientifica. 2016. https://doi.org/10.1155/2016/7097489.
-
Beck BR, Osternig LR, Oregon E. Medial tibial stress syndrome—the location of muscles in the leg in relation to symptoms. J Bone Articulation Surg Am. 1994. https://doi.org/10.2106/00004623-199407000-00015.
-
Bouche RT, Johnson CH. Medial tibial stress syndrome (tibial fasciitis)—a proposed pathomechanical model involving fascial traction. J Am Podiatr Med Assoc. 2007. https://doi.org/10.7547/0970031.
-
Beck BR. Tibial stress injuries—an aetiological review for the purposes of guiding management. Sports Med. 1998. https://doi.org/ten.2165/00007256-199826040-00005.
-
Batt ME, Ugalde V, Anderson MW, Shelton DK. A prospective controlled study of diagnostic imaging for acute shin splints. Med Sci Sports Exerc. 1998. https://doi.org/ten.1097/00005768-199811000-00002.
-
Gaeta Chiliad, Minutoli F, Scribano Eastward, Ascenti Grand, Vinci S, Bruschetta D, et al. CT and MR imaging findings in athletes with early tibial stress injuries: comparison with bone scintigraphy findings and emphasis on cortical abnormalities. Radiology. 2005. https://doi.org/10.1148/radiol.2352040406.
-
Fredericson M, Bergman AG, Hoffman KL, Dillingham MS. Tibial stress reaction in runners—correlation of clinical symptoms and scintigraphy with a new magnetic resonance imaging grading organisation. Am J Sport Med. 1995. https://doi.org/10.1177/036354659502300418.
-
Elias J. Case written report: medial tibial stress syndrome. SportEX Med. 2012;51:7–14.
-
Bartlett R, Wheat J, Robins Grand. Is movement variability of import for sports biomechanists? Sport Biomech. 2007. https://doi.org/ten.1080/14763140701322994.
-
Stergiou N, Harbourne R, Cavanaugh J. Optimal movement variability: a new theoretical perspective for neurologic physical therapy. J Neurol Phys Ther. 2006. https://doi.org/10.1097/01.npt.0000281949.48193.d9.
-
Jordan K, Challis JH, Newell KM. Walking speed influences on gait cycle variability. Gait Posture. 2007. https://doi.org/10.1016/j.gaitpost.2006.08.010.
-
Hamill J, van Emmerik REA, Heiderscheit BC, Li Fifty. A dynamical systems approach to lower extremity running injuries. Clin Biomech. 1999. https://doi.org/ten.1016/s0268-0033(98)90092-4.
Acknowledgements
Not applicable.
Funding
No funding was provided to support the preparation of this manuscript.
Author information
Affiliations
Contributions
JM conceived the idea for the review, collected and analysed the research literature, and wrote the offset full typhoon of the manuscript. KJM assisted in review and interpretation of literature, as well as reviewing all manuscript drafts. JRS was the guarantor for the review and helped with analysis and interpretation of the literature and reviewed all manuscript drafts. All authors read and canonical the last manuscript.
Corresponding author
Ethics declarations
Ideals Blessing and Consent to Participate
Not applicable.
Consent for Publication
Non applicable.
Competing Interests
Joshua Mattock, Julie Steele and Karen Mickle declare they have no competing interests relevant to the content of this article.
Boosted information
Publisher'south Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Data
Rights and permissions
Open Access This article is licensed nether a Creative Commons Attribution 4.0 International License, which permits utilize, sharing, adaptation, distribution and reproduction in whatsoever medium or format, every bit long as y'all requite appropriate credit to the original writer(s) and the source, provide a link to the Artistic Eatables licence, and indicate if changes were made. The images or other third party material in this article are included in the commodity'southward Creative Eatables licence, unless indicated otherwise in a credit line to the material. If cloth is not included in the article's Artistic Commons licence and your intended utilise is non permitted by statutory regulation or exceeds the permitted utilise, you lot will need to obtain permission directly from the copyright holder. To view a re-create of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
Nigh this commodity
Cite this article
Mattock, J.P.M., Steele, J.R. & Mickle, 1000.J. Are Leg Muscle, Tendon and Functional Characteristics Associated with Medial Tibial Stress Syndrome? A Systematic Review. Sports Med - Open 7, 71 (2021). https://doi.org/x.1186/s40798-021-00362-2
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/x.1186/s40798-021-00362-2
Keywords
- Shin splints
- Injury prevention
- Injury rehabilitation
- Leg girth
Source: https://sportsmedicine-open.springeropen.com/articles/10.1186/s40798-021-00362-2
0 Response to "Relationship of Hip Muscle Performance to Leg Ankle and Foot Injuries a Systematic Review"
Post a Comment